Abstract
Dietary calcium intake and risk of fracture and osteoporosis: prospective longitudinal cohort study.
To investigate associations between long term dietary intake of calcium and risk of fracture of any type, hip fractures, and osteoporosis.
A longitudinal and prospective cohort study, based on the Swedish Mammography Cohort, including a subcohort, the Swedish Mammography Cohort Clinical.
A population based cohort in Sweden established in 1987.
61,433 women (born between 1914 and 1948) were followed up for 19 years. 5022 of these women participated in the subcohort.
Primary outcome measures were incident fractures of any type and hip fractures, which were identified from registry data. Secondary outcome was osteoporosis diagnosed by dual energy x ray absorptiometry in the subcohort. Diet was assessed by repeated food frequency questionnaires.
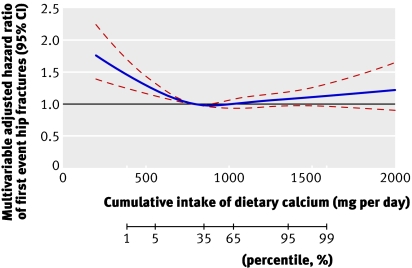
During follow-up, 14,738 women (24%) experienced a first fracture of any type and among them 3871 (6%) a first hip fracture. Of the 5022 women in the subcohort, 1012 (20%) were measured as osteoporotic. The risk patterns with dietary calcium were non-linear. The crude rate of a first fracture of any type was 17.2/1000 person years at risk in the lowest quintile of calcium intake, and 14.0/1000 person years at risk in the third quintile, corresponding to a multivariable adjusted hazard ratio of 1.18 (95% confidence interval 1.12 to 1.25). The hazard ratio for a first hip fracture was 1.29 (1.17 to 1.43) and the odds ratio for osteoporosis was 1.47 (1.09 to 2.00). With a low vitamin D intake, the rate of fracture in the first calcium quintile was more pronounced. The highest quintile of calcium intake did not further reduce the risk of fractures of any type, or of osteoporosis, but was associated with a higher rate of hip fracture, hazard ratio 1.19 (1.06 to 1.32).
Gradual increases in dietary calcium intake above the first quintile in our female population were not associated with further reductions in fracture risk or osteoporosis.
Warensjö E, Byberg L, Melhus H, Gedeborg R…
BMJ 2011
PMID: 21610048 | Free Full Text
From the full text:
• Dietary calcium intakes below approximately 700 mg per day in women were associated with an increased risk of hip fracture, any fracture, and of osteoporosis
• The highest reported calcium intake did not further reduce the risk of fractures of any type, or of osteoporosis, but was associated with a higher rate of hip fracture